-
PROVIDERS
Read more
Browse our latest selection of abstracts, manuscripts, and presentations.
-
LIFE SCIENCES
LIVE WEBINAR
Optimizing research with RWD: How Lens accelerates time to insights in drug discovery.
Register now. -
PATIENTS
It's About Time
View the Tempus vision.
- RESOURCES
-
ABOUT US
View Job Postings
We’re looking for people who can change the world.
12/17/2021
Reflections on the 2021 SABCS: harnessing the power of data to advance Breast Cancer research
Authors
Dr. Kimberly Blackwell, Tempus Chief Medical Officer
Last week, I joined thousands of in-person and virtual attendees for the 43rd annual San Antonio Breast Cancer Symposium (SABCS). From oncologists to chief medical officers to researchers to patient advocates, participants from more than 80 countries gathered to advance our clinical understanding of breast cancer, further our progress on future therapeutics, and ultimately improve patient care.
At SABCS, Tempus presented six posters that highlighted the impact of comprehensive genomic data and smart diagnostics, enabling clinicians to deliver personalized treatments for patients with breast cancer, while also powering research for future therapeutics. Conducted with researchers from Rush University Medical Center, Columbia University Irving Medical Center, Washington University School of Medicine, Northwestern University Feinberg School of Medicine, and The University of Texas Southwestern Medical Center, the presented findings underscored the value of collaboration between the private sector and academic medical centers to further personalized healthcare.
As I reflect on my four days at the symposium, here are the key takeaways from some of the most impactful presentations I had the opportunity to attend:
Value of NGS Testing in Breast Cancer: SAFIR02-Breast
- SAFIR02-Breast was a randomized prospective trial evaluating the utility of next generation sequencing (NGS) in patients with metastatic breast cancer. Following genomic profiling, patients with one of 9 actionable gene alterations were randomized to either the corresponding targeted therapies matched to genomic results, or maintenance chemotherapy. For therapeutic matches supported by the ESMO scale for clinical actionability of molecular targets (ESCAT) level I and II evidence, improved outcomes (mPFS 9.1 mo vs 2.8 mo; HR 0.41) were seen in the matched targeted therapies group. This study emphasizes the benefits of having a validated evidence-based variant calling platform, such as the MSKCC OncoKB which is used to power therapy associations in our genomic profiling reports.
ESR1 Mutations in ctDNA may signal greater benefit from Oral SERD: EMERALD Trial
- We saw the first report of the first Phase 3 trial of a novel estrogen receptor (ER) targeting drug in the EMERALD study. HR+ metastatic patients progressing on 1-2 lines of endocrine therapy including CDK 4/6 inhibitors were randomized to receive either elacestrant (an oral selective estrogen receptor degrader [SERD]) or standard of care (SOC) endocrine therapy. The presence of ESR1 mutations was identified by ctDNA analysis. Elacestrant was associated with 30% and 45% reduction in the risk of progression or death in all patients and patients harboring mESR1, respectively. This trial showed the value of circulating tumor DNA (ctDNA) for ESR1m detection to potentially determine benefit from an oral SERD. This trial took over two years to accrue a fairly common, non-selected group of patients with hormone sensitive breast cancer. While the pre-defined trial endpoints were met, further studies will be needed to clarify which patients benefit from elacestrant in second-line endocrine therapy.
Benefits from CDK 4/6 Inhibitors may differ by Intrinsic Subtypes: MONALEESA-2, -3, and -7 Subgroup Analyses
- Ribociclib added to endocrine therapy demonstrated statistically significant progression- free survival (PFS) and overall survival (OS) benefit in patients with HR+/HER- advanced breast cancer in three phase 3 clinical trials, MONALEESA-2, -3, and -7 . This retrospective analysis evaluated the association of PAM50-based intrinsic subtypes with OS. In intrinsic subtype analysis, ribociclib benefit was seen in Luminal A/B and HER2-Enriched but not in Basal types. Even with thousands of patients, the analysis of the five subgroups of ER+ breast cancer was still underpowered to make definitive conclusions, emphasizing the need for larger numbers of patients to draw conclusions about therapeutic benefit.
Supporting Evidence on Value of Monitoring ctDNA for ESR1 Mutations: PADA-1 Trial
- The PADA-1 study illustrated the value of ctDNA in monitoring HR+/HER- metastatic breast cancer patients for the presence of alterations in the ESR1 gene. Patients with no prior systemic therapy were treated with an aromatase inhibitor (AI)+palbociclib and randomized to switch to a SERD (fulvestrant+palbociclib) or continue on AI+palbociclib at the time of emergence of an ESR1 mutation. Patients who switched achieved mPFS 11.9 mo vs 5.7 mo (HR 0.63). This is the first demonstration in breast cancer regarding how sequential testing could be used to drive improved timing of switches in therapy. This same strategy is being used for randomizing patients in SERENA-6, a TIME trial supported by AstraZeneca, in which participating patients will receive routine monitoring to determine which ER mutations emerge during first line treatment.
When we convene as a group for conferences like SABCS, I’m reinvigorated with hope as this community tirelessly works towards advancing treatment options and care for patients with breast cancer.
-
04/16/2024
Reduce time to launch with Tempus and multimodal data
How AstraZeneca simplified its approach to TROPION-Lung01 and drove down time to launch.
Read more -
04/16/2024
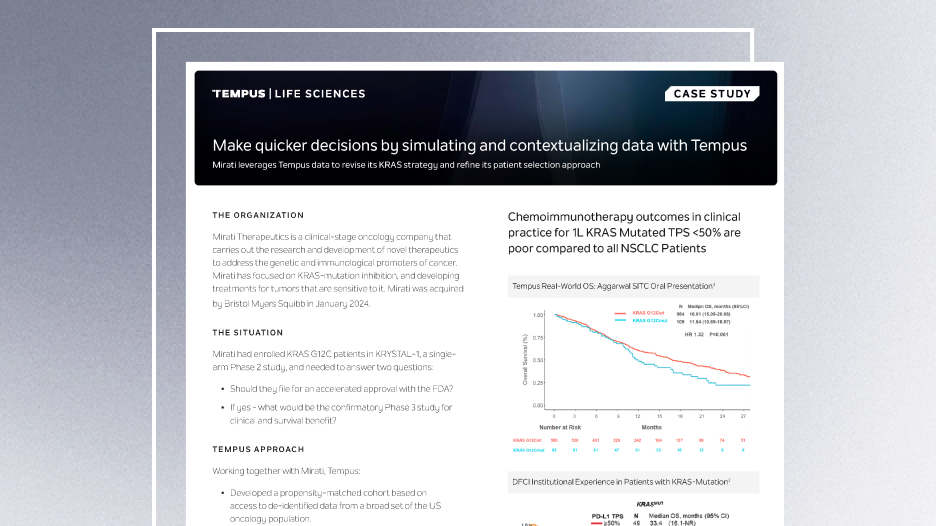
Make quicker decisions by simulating and contextualizing data with Tempus
Mirati leverages Tempus data to revise its KRAS strategy and refine its patient selection approach
Read more -
04/16/2024
Enhance your clinical development process with Tempus’ CDx platform
Learn how A2 Biotherapeutics is making progress on a breakthrough cell therapy using HLA-LOH (human leukocyte antigen-loss of heterozygosity) as a biomarker, using Tempus’ comprehensive immunotherapy platform.
Read more