-
PROVIDERS
REGISTER NOW
Upcoming Webinar:
MRD data insights across the cancer care continuum -
LIFE SCIENCES
Read now
Accelerating a phase 1 oncology trial: The TIME Network's impact on patient enrollment
-
PATIENTS
It's About Time
View the Tempus vision.
- RESOURCES
-
ABOUT US
View Job Postings
We’re looking for people who can change the world.
- INVESTORS
04/14/2026
The landmark journey of Paige Prostate: IVDR approval and building on a legacy of regulatory excellence as the first FDA-authorized AI
The journey to integrate artificial intelligence into clinical practice is defined by milestones that build trust and establish new standards of care. In 2025, Tempus achieved such a milestone: Paige Prostate received EU IVDR certification.
Authors
Razik Yousfi
SVP & GM, Imaging, Tempus

Juan Retamero
Medical VP, Pathology Operations for Diagnostic Products, Tempus

SVP & GM, Imaging, Tempus

Juan Retamero
Medical VP, Pathology Operations for Diagnostic Products, Tempus

The journey to integrate artificial intelligence into clinical practice is defined by milestones that build trust and establish new standards of care. In 2025, Tempus achieved such a milestone: Paige Prostate, including Paige Prostate Detect, Paige Prostate Grade & Quantify*, and Paige Prostate Perineural Invasion*, received EU IVDR certification.1 This achievement not only sets a new global benchmark for AI in pathology but also continues to build and expand on a legacy of regulatory leadership that began with the first-ever FDA marketing authorization for a software algorithm device to assist users in digital pathology in 2021.
For our partners in life sciences and for providers in clinical practice, these authorizations from two of the world’s most rigorous regulatory bodies offer more than a promise of innovation. They provide assurance of safety and performance, built on validation and real-world application in accordance with FDA and EU IVDR requirements.
A new global benchmark: The significance of IVDR certification
The European Union’s In Vitro Diagnostic Regulation (IVDR) represents a significant increase in regulatory scrutiny compared to the previous In Vitro Diagnostic Directive (IVDD). It demands more extensive clinical evidence, greater transparency, and a life-cycle approach to performance and safety monitoring. Achieving IVDR certification is a testament to the robustness and quality of a diagnostic tool.
By meeting these stringent requirements, Paige Prostate has demonstrated that its performance is not only clinically effective but also consistently reliable and safe for widespread use. This milestone ensures that pathologists across Europe can confidently leverage our advanced AI to enable more precise and standardized cancer diagnostics, backed by the highest international quality standards.
A legacy of regulatory firsts: Building on the landmark FDA clearance
Our journey to set the standard for AI in pathology began years ago. In 2021, Paige Prostate Detect made history by receiving the first-ever FDA De Novo marketing authorization for a software algorithm device to assist users in digital pathology. This watershed moment established a new category for digital diagnostics in the United States.
While others are now entering the field, Tempus Paige has spent the last half-decade building on this foundation. The path from the initial FDA landmark to the new IVDR certification demonstrates a sustained commitment to regulatory excellence and a proven track record that is unmatched in the industry.
The clinical evidence behind approvals
To bridge the gap between a promising AI model and a trusted clinical tool, Paige originally designed a rigorous study2 to measure the impact of its AI on pathologist performance, the results of which were published in the Archives of Pathology & Laboratory Medicine. The study simulated a real-world diagnostic workflow, where pathologists reviewed prostate biopsies first without and then with the assistance of Paige Prostate Detect.
The results provided the evidence needed to usher in a new era of AI-augmented diagnosis:
- A 70% reduction in cancer detection errors: Pathologists demonstrated a significant increase in diagnostic accuracy when assisted by Paige Prostate Detect.2
- Increased sensitivity: The ability to correctly identify cancer rose from 88.7% to 96.6%, helping to ensure that even minute cancerous foci are not overlooked.
- Improved specificity: The ability to correctly identify benign tissue also improved, which is critical for reducing false-positive diagnoses and preventing unnecessary procedures.
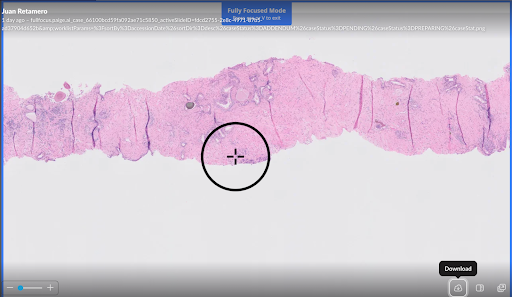
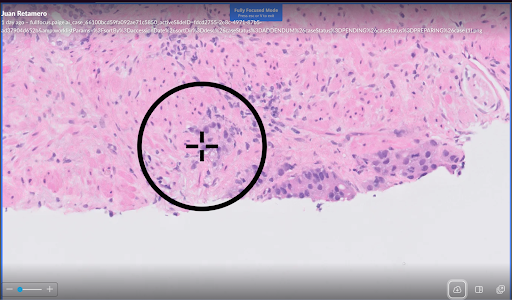
- Proven impact on challenging cases: In one powerful example, a 0.2 mm, high-grade lesion initially missed by 12 of 16 pathologists was correctly identified by all 16 with assistance from Paige’s AI application.
This powerful clinical data is the foundation of our regulatory success and the reason pathologists can trust Paige Prostate Detect as an adjunctive tool to make more informed and confident diagnoses.
Diagnostic errors reduced by 70%
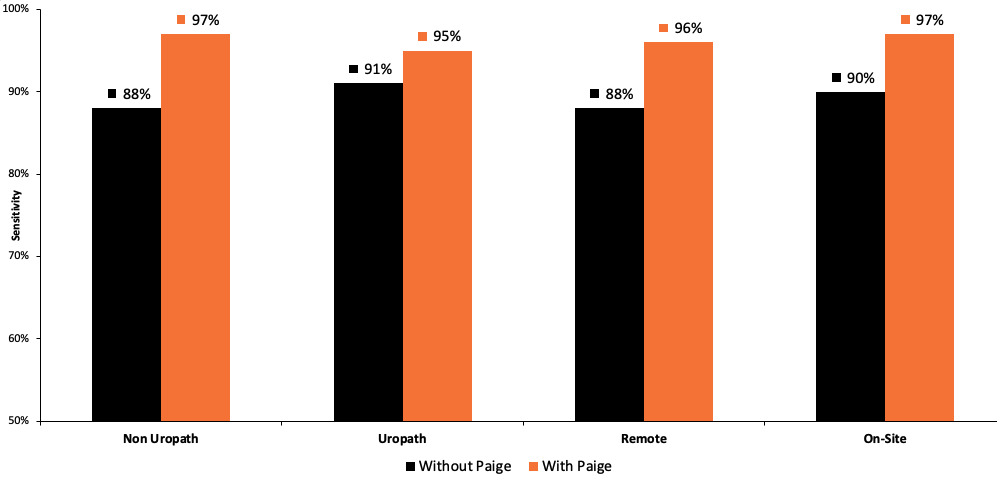
All improvements were statistically significant.
Example of pathologist false negative:
- 0.2 mm lesion
- Gleason Grade Group 4
- Misclassified as benign by 12/16 pathologists
- All pathologists arrived at correct diagnosis with Paige Prostate
Validating clinical value through partner publications
Following its original landmark FDA clearance, Paige Prostate is demonstrating significant, real-world clinical value, a fact underscored by recent publications from our partners. One publication shows how the AI model improved laboratory workflow efficiency. By acting as a reliable second opinion, it reduced requests for ancillary studies like immunohistochemistry (IHC) and shortened diagnostic turnaround times, allowing clinical teams to make critical treatment decisions more quickly.3
In another publication, Paige Prostate was shown to enhance pathologist diagnostic accuracy. It served to systematically screen slides to help pathologists mitigate the risk of false negative errors.4 Finally, in another study, Paige Prostate helped reduce the rate of equivocal diagnoses like Atypical Small Acinar Proliferation (ASAP).5 Expanding into markets outside the US, with an extended intended use, Paige Prostate also improved the correlation between biopsy and prostatectomy Gleason scores in one study, which enables more precise patient risk stratification.6
This provides pathologists with greater diagnostic certainty and can spare patients from the cost and anxiety of invasive repeat biopsies.5 Collectively, these studies provide tangible proof points for how Paige Prostate is enhancing diagnostic workflows by significantly boosting diagnostic accuracy and operational efficiency while reducing medical errors. This results in faster turnaround times and more precise treatment plans, ultimately elevating the overall patient experience.
A blueprint for the future of AI-enabled care
The journey of Paige Prostate serves as a powerful proof point for Tempus’ core mission: to build the operating system for cancer care. By structuring and analyzing vast amounts of multimodal data, we make it accessible and useful for improving patient outcomes.
As a Tempus company, Paige is an integral part of our intelligent diagnostics platform. Our unrivaled regulatory expertise extends across our entire portfolio, which includes FDA-cleared and IVD-certified tools like our xT CDx and xR IVD assays. For our pharmaceutical partners, this robust track record provides a clear and de-risked pathway to integrate regulatory-cleared AI into drug development and companion diagnostic strategies.
The story of Paige Prostate proves that we can augment the irreplaceable expertise of physicians with powerful AI to create a new standard of care. This landmark achievement is just the beginning, and at Tempus, we are proud to lead the way in building this data-driven future for all of medicine.
To learn how our globally validated AI solutions can be integrated into your clinical practice or drug development program, contact us today.
Footnotes
* For Research Use Only (RUO) in the US. Not for use in diagnostic procedures in the US.
1. CE-IVDR for use with whole slide images visualized with the Paige FullFocus Image Viewer and digitized with Philips Ultra Fast Scanner, Leica Aperio AT2 Scanner, Leica GT450 Scanner, or Hamamatsu S360 Scanner. In the US, Paige Prostate Detect is FDA-authorized for clinical use.
2. Raciti P, Sue J, Retamero JA, et al. Clinical validation of artificial intelligence–augmented pathology diagnosis demonstrates significant gains in diagnostic accuracy in prostate cancer detection. Archives of Pathology & Laboratory Medicine. 2023;147:1178-1185. doi:10.5858/arpa.2022-0303-OA. https://pubmed.ncbi.nlm.nih.gov/36538386/
3. Eloy C, Marques A, Pinto J, et al. Artificial intelligence–assisted cancer diagnosis improves the efficiency of pathologists in prostatic biopsies. Virchows Archiv. 2023;482:595-604. doi:10.1007/s00428-023-03518-5. https://link.springer.com/article/10.1007/s00428-023-03518-5
4. da Silva LM, Pereira EM, Salles PG, et al. Independent real-world application of a clinical-grade automated prostate cancer detection system. The Journal of Pathology. 2021;254:147-158. doi:10.1002/path.5662. https://pubmed.ncbi.nlm.nih.gov/33904171/
5. Liu S, Jamal M, Smotherman C, et al. The potential use of artificial intelligence in reviewing atypical small acinar proliferation in prostate core biopsy. Virchows Archiv. 2026;488:761-766. doi:10.1007/s00428-025-04136-z. https://link.springer.com/article/10.1007/s00428-025-04136-z
6. Eloy C, Asaturova A, Pinto J, et al. Clinical advantages in providing artificial intelligence-assisted prostate cancer diagnosis: A pilot study. Pathology – Research and Practice. 2025;271:156007. doi:10.1016/j.prp.2025.156007. https://www.sciencedirect.com/science/article/pii/S0344033825001992
-
03/04/2026
Supporting the financial health of clinical research programs with the Tempus TIME Trial Network
By combining the power of AI-driven technology with a streamlined operational model, Tempus provides the infrastructure and support to enable sites with limited resources to participate in research and expand their capabilities.
Read more -
01/20/2026
From image to insight: How Tempus is leveraging AI to advance biomarker screening at scale
By leveraging Paige Predict to screen for genomic and phenotypic biomarkers, clinicians and researchers have rapid access to valuable information to prioritize testing and pre-screen patients at scale.
Read more -
12/04/2025
Pioneering decentralized oncology trials: Success at the nation’s largest community practices
Hear directly from industry leaders and pioneering site partners as they share insights on expanding their research footprint and improving financial sustainability for their institutions.
Watch replay


