-
PROVIDERS
Learn more
Join Tempus at the 2026 ASCO® Annual Meeting!
-
LIFE SCIENCES
Read case study
Learn now Tempus Next Pathways unlocked critical insights into the aNSCLC patient journey with Bristol Myers Squibb (BMS)
-
PATIENTS
It's About Time
View the Tempus vision.
- RESOURCES
-
ABOUT US
View Job Postings
We’re looking for people who can change the world.
- INVESTORS
BIOLOGICAL MODELING
Our vast tumor organoid repository enables new possibilities for therapy selection and drug discovery
We aim to transform therapy selection and drug discovery and development by building one of the world’s largest libraries of human ex vivo tumor-derived organoids that are characterized by molecular features and associated clinical outcomes in robust, reproducible 3D models.
Contact usTempus’ Patient-Derived Organoid (PDO) Screens
Test and rapidly assist in evaluating your preclinical drug candidates with our fixed PDO panels, designed for diverse therapeutic applications and offered on a rotating quarterly schedule.
- Pan-indication panels: Large panels featuring PDOs across numerous indications to support a wide range of research questions.
- Indication-specific panels: Focused panels enriched for models within a specific indication, such as colorectal, lung, or breast cancer.
- Biomarker-specific panels: Targeted panels designed for key mutations or biomarkers such as KRAS, PIK3CA mutations, MSI-High, etc.
View the panels currently open for enrollment and secure your spot.
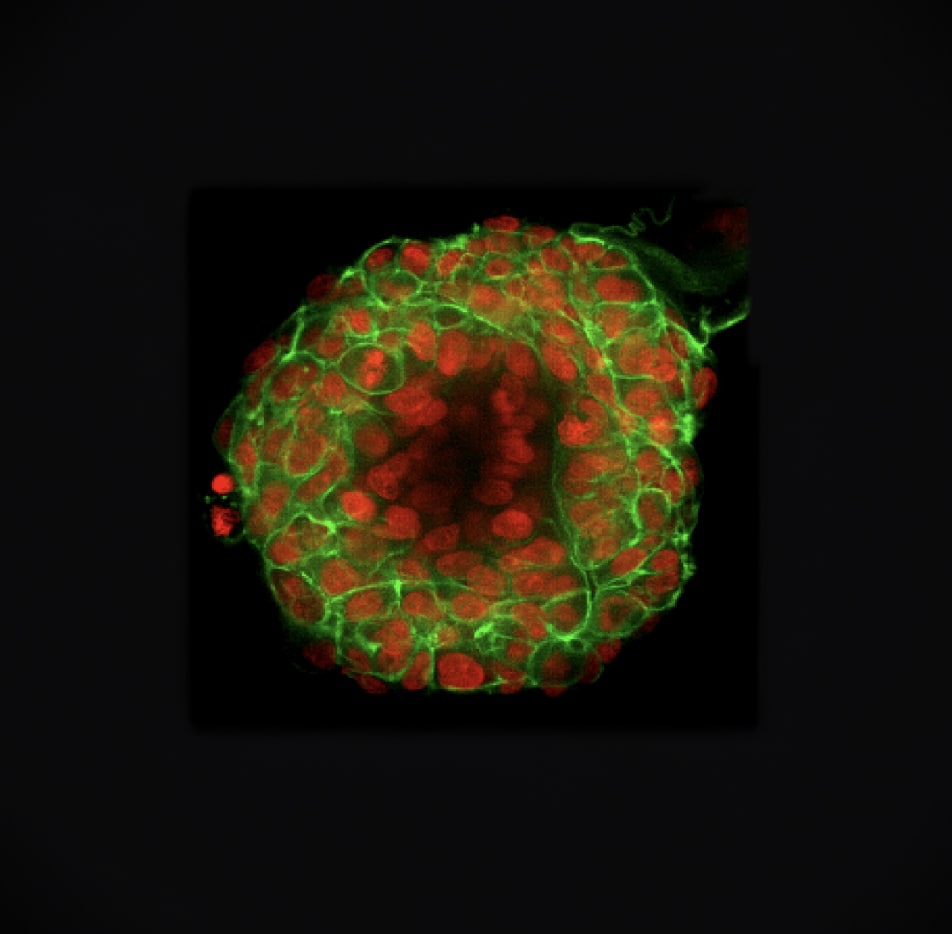 Lung cancer TO 3D morphology, with fluorescent staining of nuclei and cytoskeleton.
Lung cancer TO 3D morphology, with fluorescent staining of nuclei and cytoskeleton.
Our Solutions
Predefined organoid screening models
Predefined sets of organoid models enriched for specific characteristics (e.g., indication, biomarker, clinical data) can be used to screen test compounds and to test and generate hypotheses. Compounds can be screened alone and in combinations, providing a rapid and cost-effective alternative to in vivo preclinical animal experiments.
Learn more about our fixed panelsCustom organoid screening models
Client-selected organoids screened with or without allogeneic MHC-matched PBMCs, tailored to your research needs. A variety of compound classes can be screened across small molecules, mAbs, ADCs, cell engagers, and cellular therapies.
- Tumor Origin: Pan-Cancer, client-selected indications
- Molecular Profiling: Client-selected molecular alterations, mutations, CNVs, fusions, etc.
Single-cell RNA sequencing
Characterize cell populations of interest with precision–identify and validate complex signatures and identify biomarkers of therapeutic response
Case study
A large Biopharma collaborator used Tempus’ patient-derived organoids to unlock a critical investment decision. Read More
PAN-CANCER TUMOR ORGANOIDS
-
Breast
-
Colon
-
Endometrial Adeno
-
Gastric
-
Head + Neck SCC
-
Liver HCC
-
NSCLC Adeno
-
NSCLC SCC
-
Ovarian
-
Pancreatic
Our solutions can be broadly applied to preclinical and post-approval strategies
- Biomarker and target validation
- Indication selection
- Exploratory (MOA) studies
- Exploration of rare patient mutations and alterations
- Combination identification and validation
- RNA signature development for responder enrichment
- Therapy resistant models to study disease biology
- Immune cell activation
- Label expansion plans
Our Differentiators
-
Full characterization
of tumor organoids with Tempus xT solid tumor comprehensive profiling
-
Flexible
3D confocal imaging assays with AI analytics on every tumor organoid drug sensitivity screen
-
Innovative pipeline
of automation-driven assays enables expedited experimental processes
-
CAP-accredited, CLIA-certified
laboratories

How it works
Individual human ex vivo tumor-derived organoids are screened in Tempus’ high-throughput environment. Tempus organoids are NGS-characterized and have been pre-screened against a panel of chemotherapies and small molecule therapeutics. Our 3D models serve as the basis for drug screening efforts for drug discovery and development.
-
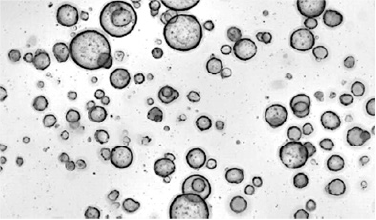
Organoids are grown in proprietary chemically defined conditions from core biopsies or surgical resections.
-
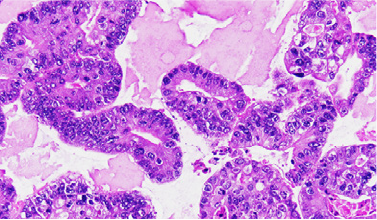
Organoid histology is verified by board-certified medical pathologists.
-
Full transcriptomic profiles are generated by RNA sequencing.
-

Tumor mutation recapitulation is assessed by overlap of somatic variants between tumor and tumor organoids via the Tempus xT & xR assays.
-
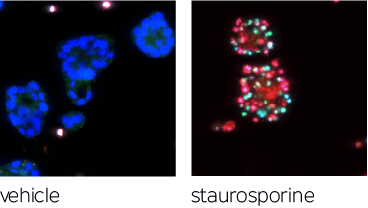
Organoids are phenotyped via high-content assays, which are then analyzed via automated machine vision algorithms employing state-of-the-art computer vision models to segment organoids, quantify infiltrating immune cells, and predict drug efficacies along with other clinical endpoints as desired.
-
Precision Oncology for High-Grade Gliomas: A Tumor Organoid Model for Adjuvant Treatment Selection
Tripathy et al. refined the generation of multiregional patient-derived GTOs across various glioma subtypes, preserving transcriptomic and phenotypic heterogeneity of aggressive HGGs. High-throughput screening revealed marked variability in drug response both between patients and across tumor regions, suggesting the necessity of multi-agent therapy and highlighting the clinical utility of multiregional GTOs for personalized treatment.
READ PUBLICATION -
Identification of Predictive Biomarkers of Response to ADCs by HTS of Highly Molecularly Characterized Panels of Patient-Derived Organoids (PDOs)
Tan et al. developed and screened a diverse panel of 60 PDOs across 10 solid tumor indications, identifying ADC-specific responses driven by target expression and uncovering potential biomarkers to enhance understanding of ADC efficacy.
READ PUBLICATION -
Stratification of Cell Therapies in Solid Tumor Organoids Using Deep Learning-Derived Imaging Metrics
Lonini et al. developed a scalable platform that utilized deep learning to predict PDO viability from over 11,000 brightfield time-lapse images, eliminating the need for fluorescent dyes and enabling dynamic response readouts for 27 cell therapies across 60 PDOs and 9 cancer types, while also extracting interpretable phenotypic features that categorized therapies into functional groups based on their biological effects.
READ PUBLICATION
Trusted by hundreds of biopharma to power drug development
The information on this page is intended for life sciences companies and focuses on research and development applications.
Partnering with Tempus is investing in the future
We’ve created a biological modeling infrastructure centered on ex vivo tumor organoids to help you accelerate your research needs.