-
PROVIDERS
REGISTER NOW
Upcoming Webinar:
MRD data insights across the cancer care continuum -
LIFE SCIENCES
REGISTER NOW
From insight to impact: Leveraging the AI-enabled Next platform with BMS to advance equitable access in precision oncology
-
PATIENTS
It's About Time
View the Tempus vision.
- RESOURCES
-
ABOUT US
View Job Postings
We’re looking for people who can change the world.
- INVESTORS
04/27/2026
MUC16 in Ovarian Cancer: High, Stable Expression Across Histologic Subtypes, Disease Stages, and Platinum Sensitivity Status Supports Antibody-Drug Conjugate Development
SGO 2026
PRESENTATION
Authors
Ursula Matulonis, Robert Coleman, Michael Birrer, Fernanda Musa, David O'Malley, Maria Rubinstein, Ritu Salani, Dayana Delgado, Lennart Langouche, David Dornan, Kathleen Keegan, David Lennon, Ashwini Pai, Kathleen Moore
Objectives
MUC16, a glycoprotein cleaved to form serum CA125, is upregulated in ovarian cancer. Early antibody-drug conjugates (ADCs) targeting MUC16 comprised antibodies targeted to the cleaved portion of MUC16 and therefore were potentially limited by serum CA125 binding acting as an antigen sink and limiting tumor access, as well as by sub-optimal linker–payload design. Advances in ADC technology have renewed interest in MUC16 as a therapeutic target. Previous analyses of MUC16 expression patterns in ovarian cancer using public databases have been limited by small samples sizes and lack of histology-specific data. This study aimed to assess MUC16 expression in a large population, across diverse histologic subtypes and stages of ovarian cancer.
Methods
Data from Clinical Proteomic Tumor Analysis Consortium were used to assess MUC16 protein abundance in tumor vs normal tissue using the Wilcoxon test, and to compare with MUC16 mRNA levels using Spearman’s correlation. RNA sequencing data (Tempus AI, Inc.) were used to examine MUC16 expression across ovarian cancer histologic subtypes, disease stages, primary vs metastatic tumors, and platinum sensitivity status. Median mRNA expression was quantified as log2(transcripts per million [TPM]+1) and differences were evaluated using the Kruskal-Wallis test.
Results
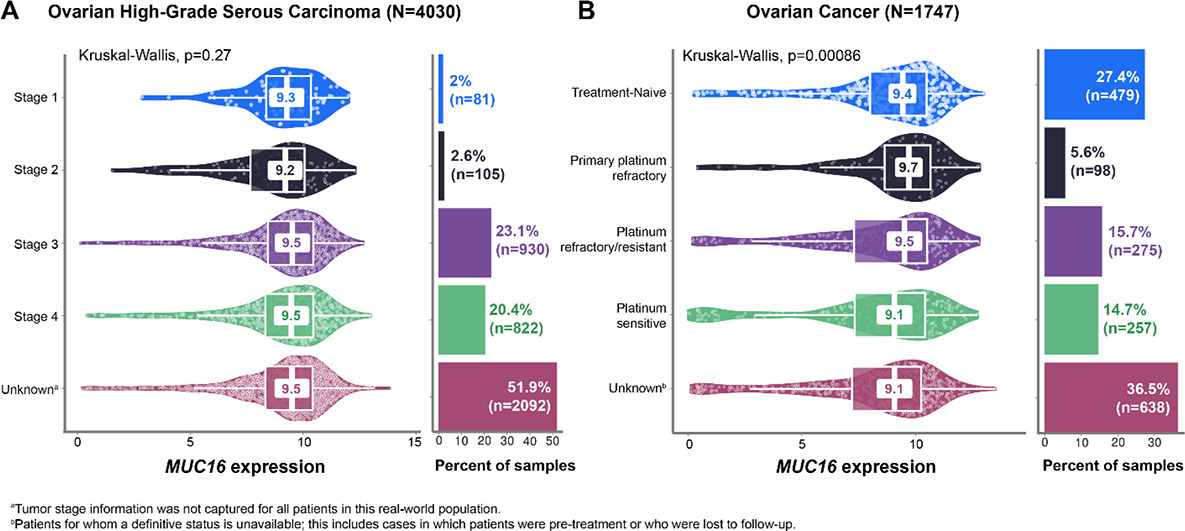
MUC16 protein abundance in ovarian serous carcinoma tumor samples was 1.35-fold greater than in normal, adjacent tissue samples and was positively correlated with MUC16 mRNA levels (Spearman’s ρ = 0.49). Across nine histologic subtypes (N = 7244), overall median MUC16 expression (log2[TPM+1]) was 9.1. Median MUC16 expression was higher than that of other ovarian ADC targets such as MSLN (8.0), SLC34A2 (NaPi2b) (7.8), ERBB2 (HER2) (7.1), FOLR1 (FRα) (7.0), TACSTD2 (Trop-2) (6.9), CDH6 (5.2), VTCN1 (B7-H4) (5.2), NECTIN4 (4.1), and CLDN6 (2.1). In high-grade serous carcinoma samples (n = 4030), median MUC16 expression was 9.5 overall and remained stable across primary vs metastatic tumors (9.5 in both) and disease stages (9.2–9.5; Fig. A). Median MUC16 expression was 8.0 in endometrioid carcinoma (n = 429), 6.3 in clear-cell adenocarcinoma (n = 381), and 6.2 in carcinosarcoma (n = 228), all non-serous subtypes. Median MUC16 expression in platinum–refractory/resistant and –sensitive tumor samples was 9.5 and 9.1, respectively (Fig. B).
Conclusions
MUC16 is potentially the most highly expressed biomarker among current therapeutic targets in ovarian cancer. While expression was lower in non-serous compared with serous tumors, MUC16 was expressed in these tumors (log2[TPM+1] 6.2–8.0) to an extent similar to or greater than other targets being developed in ovarian cancer (log2[TPM+1] 2.1–8.0). MUC16 showed tumor-selective protein enrichment and mRNA–protein concordance, and expression was stable across most histologic subtypes, disease stages, and irrespective of platinum sensitivity. Taken together, these results support MUC16 as a promising therapeutic target that warrants further investigation.