-
PROVIDERS
REGISTER NOW
Upcoming Webinar:
MRD data insights across the cancer care continuum -
LIFE SCIENCES
REGISTER NOW
From insight to impact: Leveraging the AI-enabled Next platform with BMS to advance equitable access in precision oncology
-
PATIENTS
It's About Time
View the Tempus vision.
- RESOURCES
-
ABOUT US
View Job Postings
We’re looking for people who can change the world.
- INVESTORS
04/27/2026
Genomic and Transcriptomic Analysis To Uncover Potential Biomarkers of Response and Primary Resistance to Mirvetuximab Soravtansine in Patients With Ovarian Cancer
SGO 2026
PRESENTATION
Authors
Jordyn Silverstein, Sinead Cullina, Samer Alkassis, Stamatina Fragkogianni, Unnati Jariwala, Jacob Mercer, Aditya Bardia, Gottfried Konecny
Objectives
Mirvetuximab soravtansine (MIRV), a first-in-class folate receptor alpha (FRα) targeted antibody-drug conjugate (ADC), demonstrated a survival benefit over chemotherapy in the phase III MIRASOL study. To date, no study has evaluated downstream biomarkers that may modulate response which may occur at multiple steps of the ADC pathway. Here, we investigated genomic and transcriptomic alterations that may drive response or primary resistance to MIRV.
Methods
We used Tempus Lens, a platform used to query multimodal data from millions of de-identified patient records in the Tempus Database to identify patients (pts) who had Tempus xT (DNA) or xR (RNA) testing prior to or 15 days after MIRV treatment (N = 98). We used MIRV treatment duration to classify pts as responders (MIRV-R, >9 weeks) or non-responders (MIRV-nR, <= 9 weeks). RNA-seq data were normalized and quantified as transcripts per million (TPM) and reported as log2(TPM + 1) for genes related to ADC mechanisms of action. Median gene expression was compared using the Wilcoxon rank-sum test. Real-world overall survival (rwOS) was defined as the time from MIRV start to death or loss to follow-up. Median rwOS was estimated using Kaplan Meier curves and compared using a log-rank test. Given the exploratory nature of the analysis, all values are reported as uncorrected.
Results
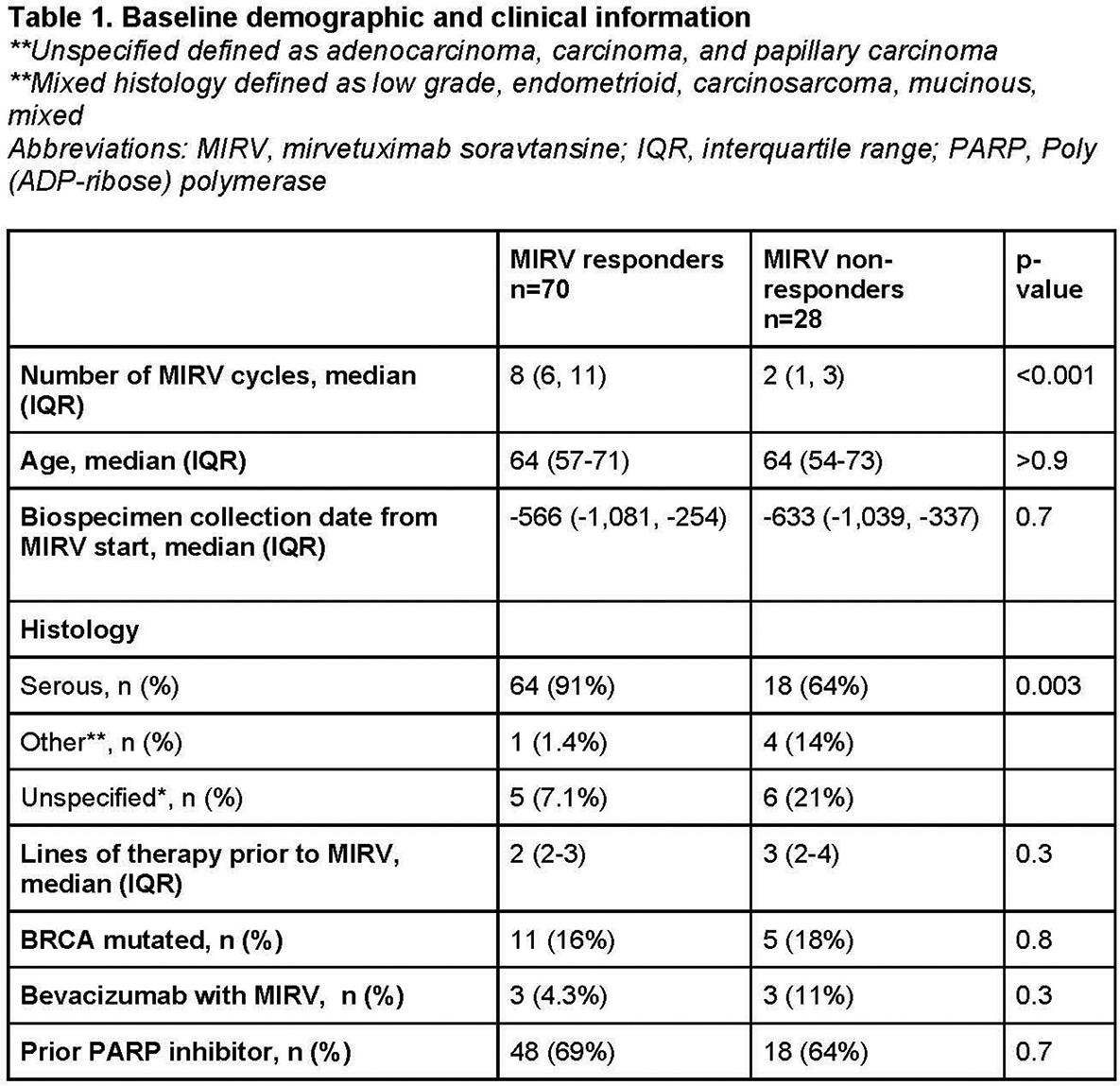
Among 98 pts, 28 (28.5%) were MIRV-nR, and 70 (71.4%) MIRV-R. Baseline demographic and clinical characteristics can be found in Table 1. MIRV-R were significantly enriched in serous ovarian cancer histologies compared to MIRV-nR pts (91% vs 64%, p = 0.003).
Pathways downstream of FOLR1 such as JAK1 (7.2 vs 7.0, p = 0.02) and MAPK1 (4.5 vs 4.3, p = 0.05) were significantly upregulated in MIRV-nR compared to MIRV-R. Expression of the drug efflux pump ABCC1 trended higher in MIRV-nR (6.7 vs 6.5, p = 0.06). No significant differences were observed in glutathione production, endolysosomal processing and known payload resistance mechanisms. Median rwOS was 14.5 months (95% confidence interval: 11.2–16.5) and significantly longer in responders compared to non-responders (16.6 vs 3.4 months, p < 0.001).
Conclusions
Our data suggest that upregulation of JAK1 and MAPK1 pathways may contribute to primary resistance to MIRV through enhanced survival and proliferation signaling. Increased ABCC1 expression may also reduce payload efficacy. Larger studies integrating genomic and transcriptomic profiling with clinical outcomes are needed to validate these findings and inform rational combination strategies to further improve efficacy with MIRV.