-
PROVIDERS
Learn more
Join Tempus at the 2026 ASCO® Annual Meeting!
-
LIFE SCIENCES
REGISTER NOW
From insight to impact: Leveraging the AI-enabled Next platform with BMS to advance equitable access in precision oncology
-
PATIENTS
It's About Time
View the Tempus vision.
- RESOURCES
-
ABOUT US
View Job Postings
We’re looking for people who can change the world.
- INVESTORS
10/16/2025
A Randomized Trial Evaluating the Impact of Automated Notifications on the Identification and Treatment of Patients With Aortic Stenosis and Mitral Regurgitation: Design of the ALERT Trial
CVCT 2025
PRESENTATION
Authors
W. Batchelor, B. Lindman, M. Coylewright, A. Keller, M. Sotelo, D. Shin, C. Rogers, G. Hickey, J. Williams, Y. Chang, S. Vemulapalli
BACKGROUND/AIMS
Severe aortic stenosis (AS) and mitral regurgitation (MR) are common valvular heart disease conditions associated with significant morbidity and mortality if not identified and treated in a timely fashion. Despite the availability of effective transcatheter and surgical therapies, many patients, especially those from minority, female, and rural populations, do not receive timely referral or intervention, contributing to persistent healthcare disparities.
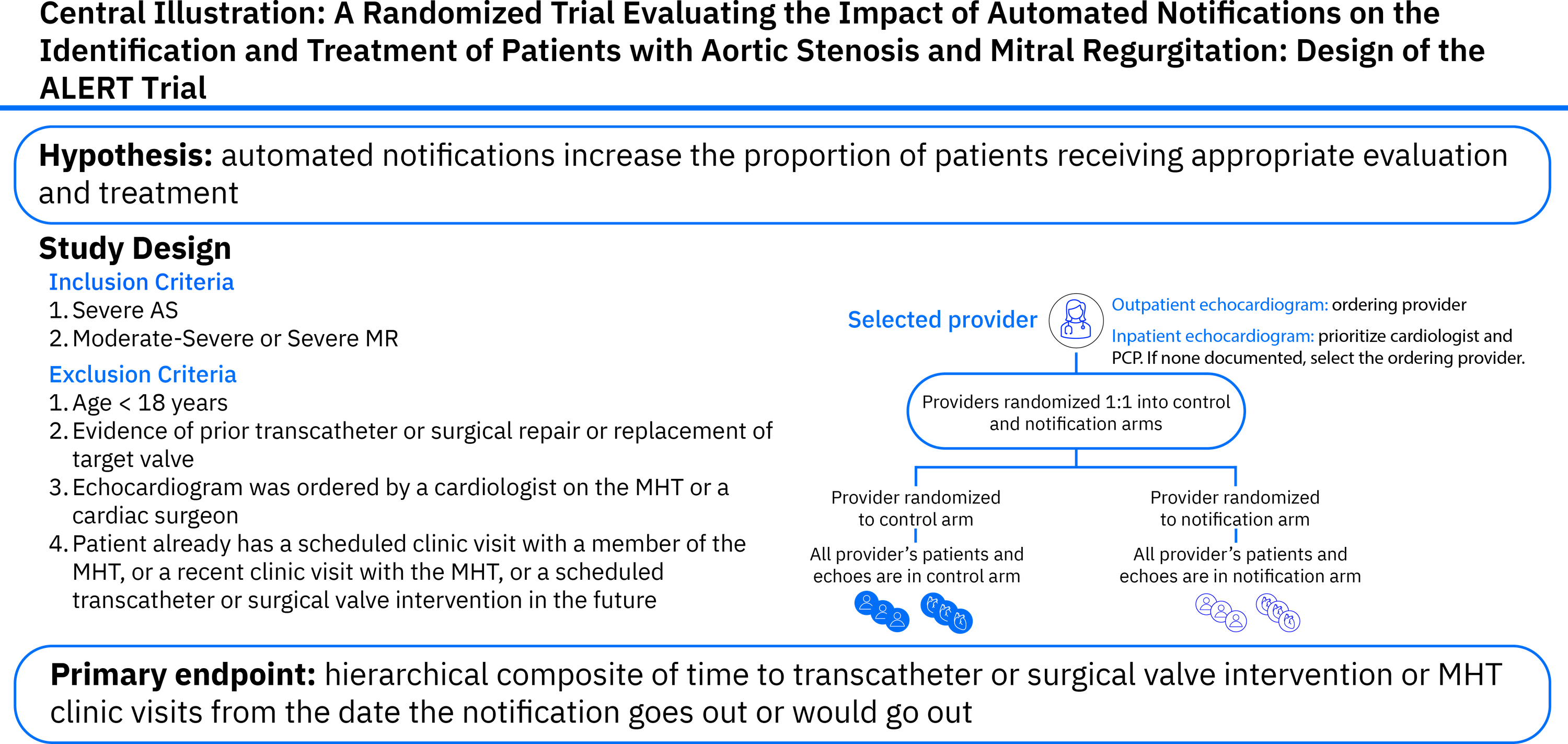
The ALERT (Addressing undertreatment and heaLth Equity in aortic stenosis and mitral regurgitation using an integrated ehR plaTform) trial aims to evaluate whether automated electronic health record (EHR) notifications to providers can improve the identification, evaluation, and treatment of patients with severe AS or moderate-to-severe/severe MR and reduce disparities in care.
METHODS
ALERT is a multi-center, prospective, cluster-randomized controlled trial enrolling patients from 5 US hospital systems. Providers ordering echocardiograms are randomized 1:1 to receive automated notifications or usual care. Notifications are triggered by echocardiogram findings consistent with severe AS or moderate-to-severe/severe MR in patients without an EHR record of a visit or clinician note by a member of the multidisciplinary heart valve team (MHT). The primary endpoint is a hierarchical composite of time to transcatheter or surgical valve intervention or time to MHT clinic visit within 90 days of enrollment. Secondary endpoints include rates of individual components of the composite. The trial aimed to randomize at least 600 providers and 1,500 patients. Data are collected via EHR integrations and analyzed using a disease stratified win ratio approach.
RESULTS
As of August 2025, enrollment has ended, with 2006 patients and 741 providers enrolled across participating sites. The trial is powered to greater than 80% at a total sample size of 1,246 patients or greater, based on the assumption that alerts will result in an absolute increase the incidence of valve intervention and MHT referral of 5% within 90 days (which entails hazard ratios between 1.5 and 2.05). Prespecified subgroup analyses will assess the impact of the intervention on healthcare disparities, including race, sex, and site.
CONCLUSION
The ALERT trial leverages an integrated EHR platform to address the undertreatment of valvular heart disease and disparities in patient care. If successful, automated notifications could represent a scalable, minimally disruptive intervention to improve quality and equity of care for patients with AS and MR.
Design of the ALERT trial