-
PROVIDERS
New MRD Medicare Coverage for Select Indications*
*When coverage criteria are met. Additional criteria and exceptions for coverage may apply.
-
LIFE SCIENCES
Read now
Accelerating a phase 1 oncology trial: The TIME Network's impact on patient enrollment
-
PATIENTS
It's About Time
View the Tempus vision.
- RESOURCES
-
ABOUT US
View Job Postings
We’re looking for people who can change the world.
- INVESTORS
11/15/2025
Multi-omic analysis of 1001 GIST tumors reveals immune enriched subtypes
CTOS 2025
PRESENTATION
Authors
Nathan D. Seligson, Nathan D. Maulding, Andrew J. Sedgewick, Danielle E. Hamel, Jason K. Sicklick, James L. Chen
Objective
Gastrointestinal stromal tumors (GIST) have occasionally responded to immunotherapy (IO), but responses are rare and unpredictable. To this end, we applied Immune Profile Score (IPS), a prognostic biomarker for IO response across multiple cancer types, to the largest cohort of multi-omic profiled GIST tumors reported to evaluate the molecular landscape to identify subsets of GIST that may benefit from IO.
Methods
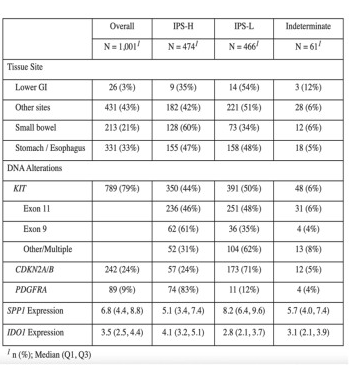
We used the Tempus multi-modal database to retrospectively select 1,001 de-identified NGS records (collected/sequenced between 2010-2024) from 926 GIST patients, each with curated clinical data. Tempus IPS is a continuous value that is divided into high (IPS-H), low (IPS-L) or indeterminate based on predefined thresholds, where IPS-H corresponds to better predicted response to IO.
Results
Patient demographics were similar to previously published cohorts: age at diagnosis (62 years), female (46%), alterations in: KIT (76%), PDGFRA (9%) and CDKN2A/B loss (24%). GISTs from primary sites were predicted to be IPS-H in 51% of cases versus 42% in distant metastases (p< 0.005). Early-stage stomach/esophageal tumors were more likely to be IPS-H than advanced-stage (60% vs 24%, p< 0.001); this effect was absent in small bowel GISTs. Across all sites, IPS-H tumors were associated with PDGFRA alterations (p< 0.001) while IPS-L tumors were enriched for CDKN2A/B loss (p< 0.001). IPS-H was more prevalent in GIST with alterations in KIT exon 9 (61%) than in KIT exon 11 (46%) or in exons other than 9 and 11 (32%). Compared to all protein coding genes, IPS model genes SPP1 and IDO1 ranked 1st and 3rd by variance of expression and have been previously linked to IO and TKI resistance.
Conclusion
In the largest multi-omic evaluation of GIST patients to date, we report clear clinical and molecular biomarkers associated with a signature of IO sensitivity. Clinical studies will be required to confirm these findings.