-
PROVIDERS
New MRD Medicare Coverage for Select Indications*
*When coverage criteria are met. Additional criteria and exceptions for coverage may apply.
-
LIFE SCIENCES
Register now
UPCOMING WEBINAR
Unlocking Foundation Models: Our experience from proof of concept to deploying at scale -
PATIENTS
It's About Time
View the Tempus vision.
- RESOURCES
-
ABOUT US
View Job Postings
We’re looking for people who can change the world.
- INVESTORS
10/14/2025
Target RNA Expression by Tumor Molecular Subtypes in Patients (Pts) With Advanced Urothelial Carcinoma (aUC) or Muscle Invasive Bladder Cancer (MIBC): Exploratory Analyses From JAVELIN Bladder 100 and the Tempus Database
ESMO 2025
Abstract
Authors
M. Eckstein, N. Klümper, P. Grivas, E. Grande, J. Brägelmann, J. Hoffman, J. Mazur, O. Bogatyrova, V. Gruenwald
Background – Molecular targets of investigational or novel therapies include Nectin-4, HER2, HER3, EGFR, and TROP2. Consensus molecular subtypes of UC/BC comprise basal/squamous (Ba/Sq), stroma-rich (SR), neuroendocrine (NE)-like, luminal unstable, luminal papillary, and luminal nonspecified. We report exploratory analyses of molecular target RNA expression by tumor subtype in pts with aUC or MIBC from the JAVELIN Bladder 100 phase 3 trial and Tempus real-world database to generate hypotheses for future studies.
Methods – JAVELIN Bladder 100 (NCT02603432) enrolled pts with aUC without progression after first-line platinum-based chemotherapy. Pts with MIBC or aUC were identified in the Tempus database of deidentified pt data from US clinical practice. Whole-transcriptome profiles in tumor samples were generated using RNA sequencing. NECTIN4, HER2, HER3, EGFR, and TROP2 transcript levels were quantified using Personalis ACE technology (JAVELIN Bladder 100) or kallisto (Tempus).
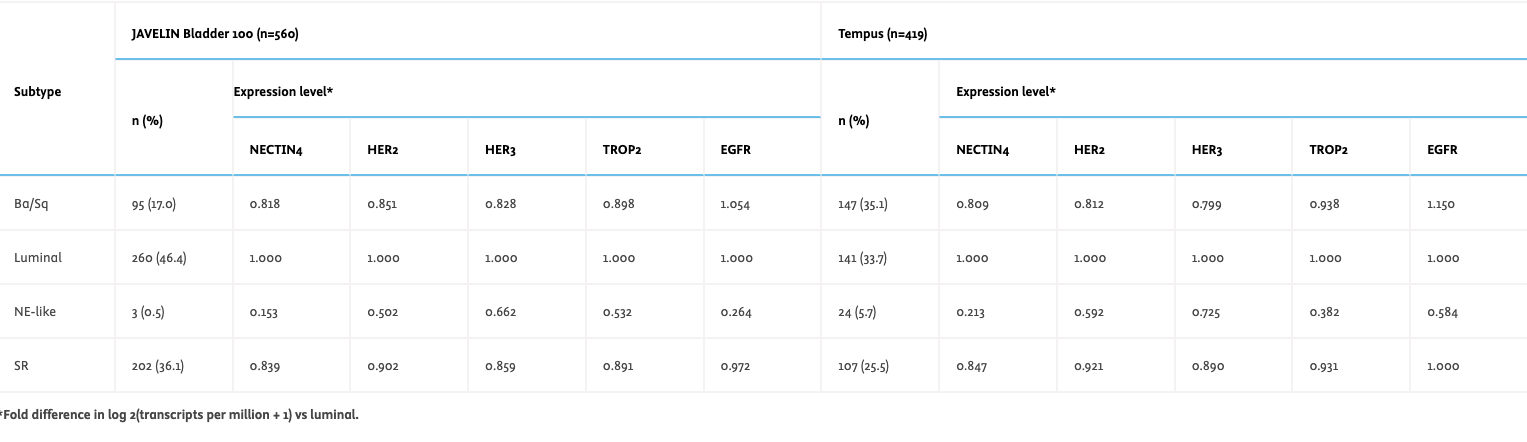
Results – In evaluable aUC tumors from the JAVELIN cohort (n=560) and MIBC tumors from the Tempus database (n=419), NECTIN4, HER2, HER3, and TROP2 RNA expression was highest in luminal subtypes followed by SR and Ba/Sq, and lowest in the NE-like subtype (Table). EGFR RNA expression was highest in Ba/Sq followed by luminal and SR, and lowest in the NE-like subtype. In both cohorts, a strong correlation (R>0.5) was observed between RNA expression of NECTIN4 and TROP2, HER2, and HER3, and a weaker correlation was observed with EGFR.
Conclusions – RNA expression patterns of various molecular targets differ between consensus molecular subtypes of UC/BC. Studies to further assess the activity of novel therapies, relevant targets, and biomarkers in consensus molecular subtypes may be warranted to individualize treatment and optimize clinical outcomes.
Clinical trial identification – NCT02603432.