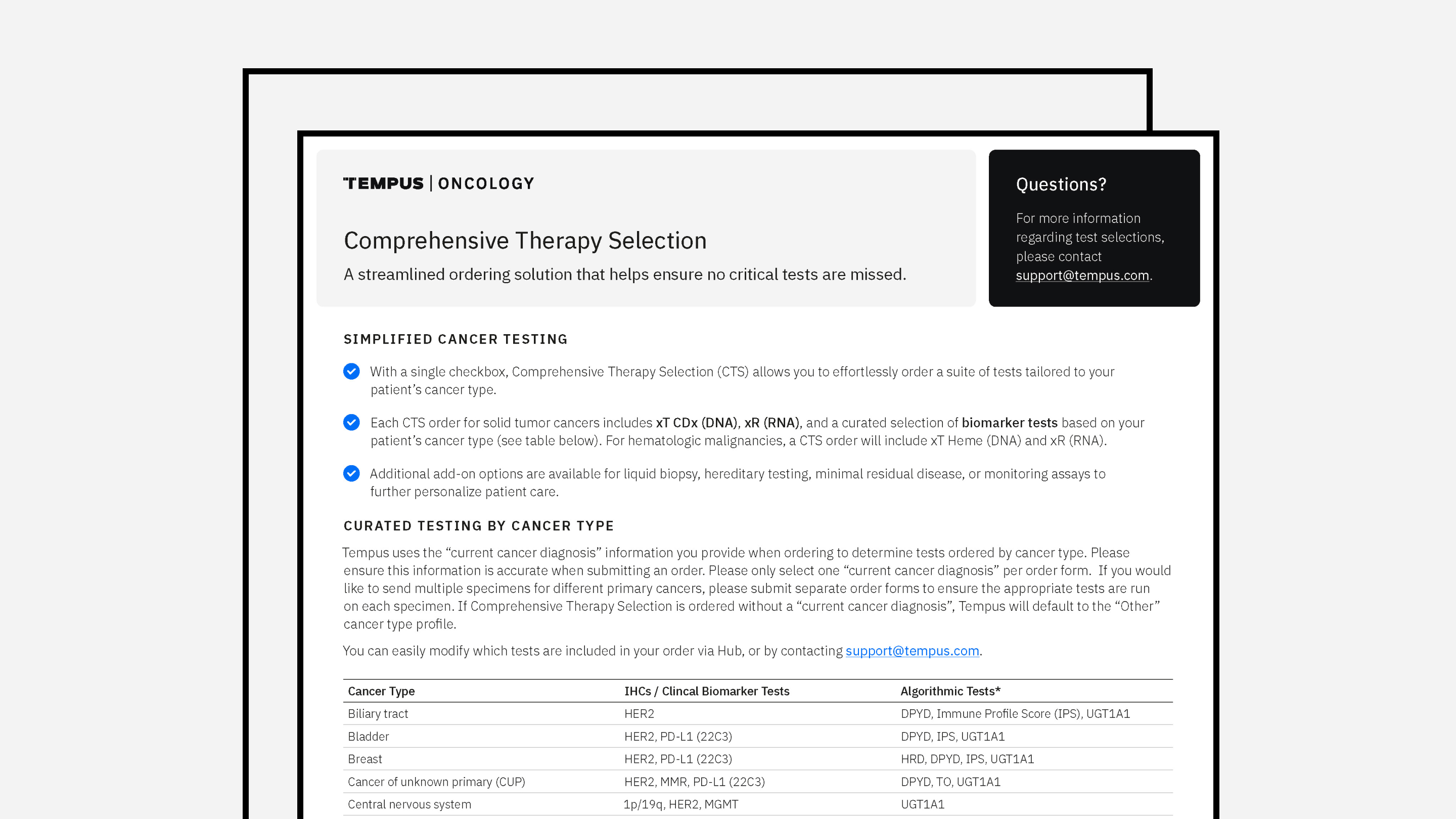
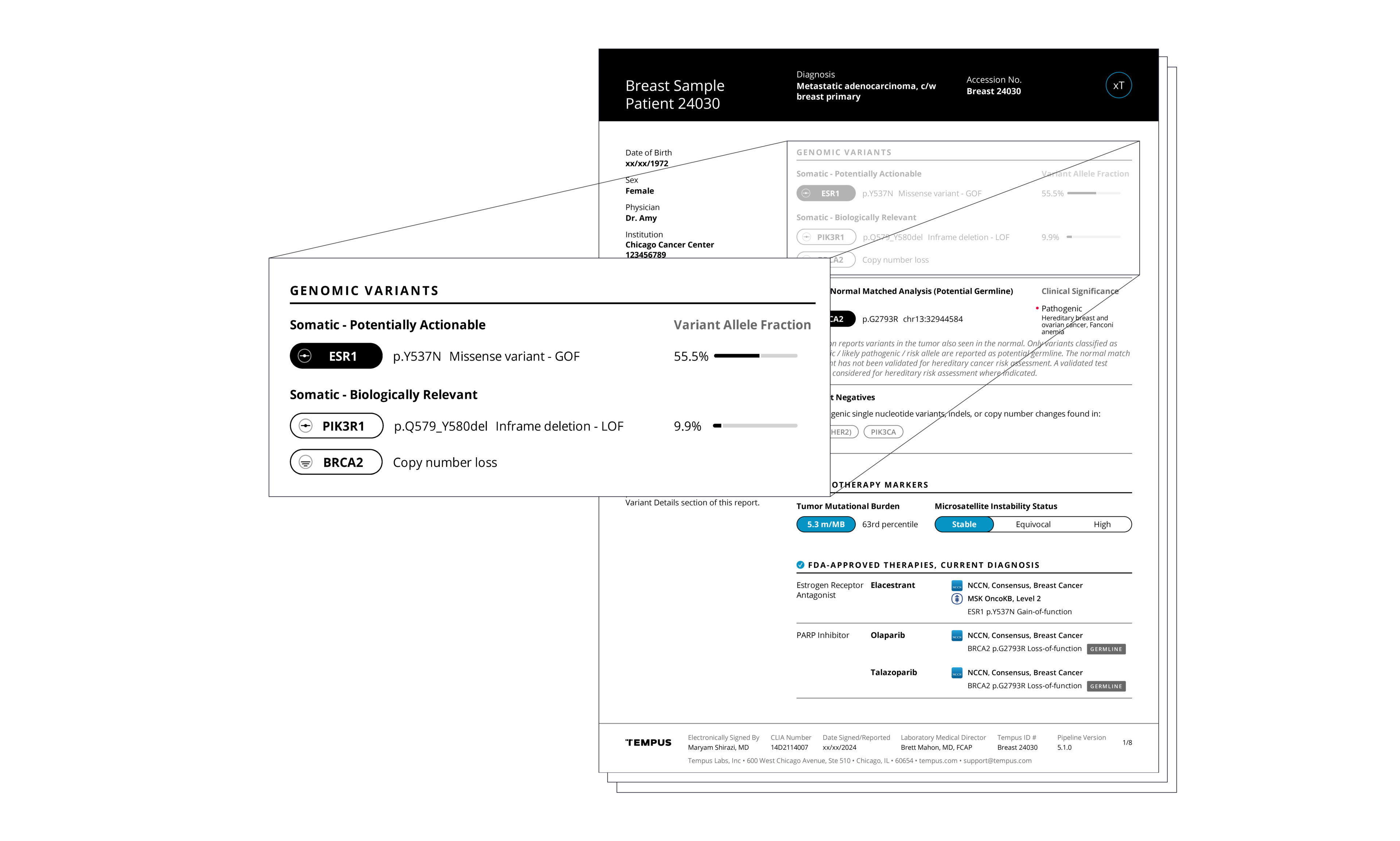
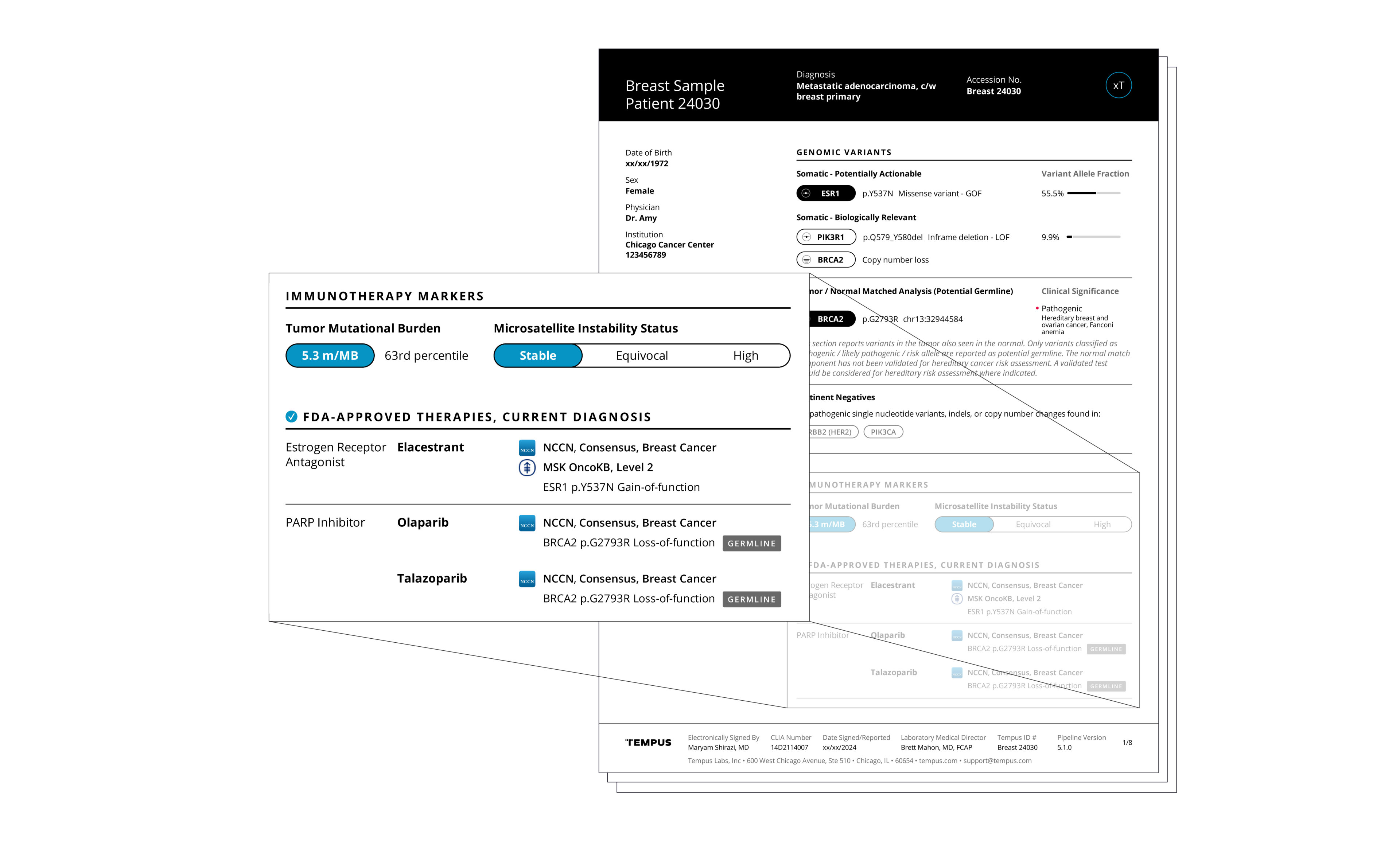
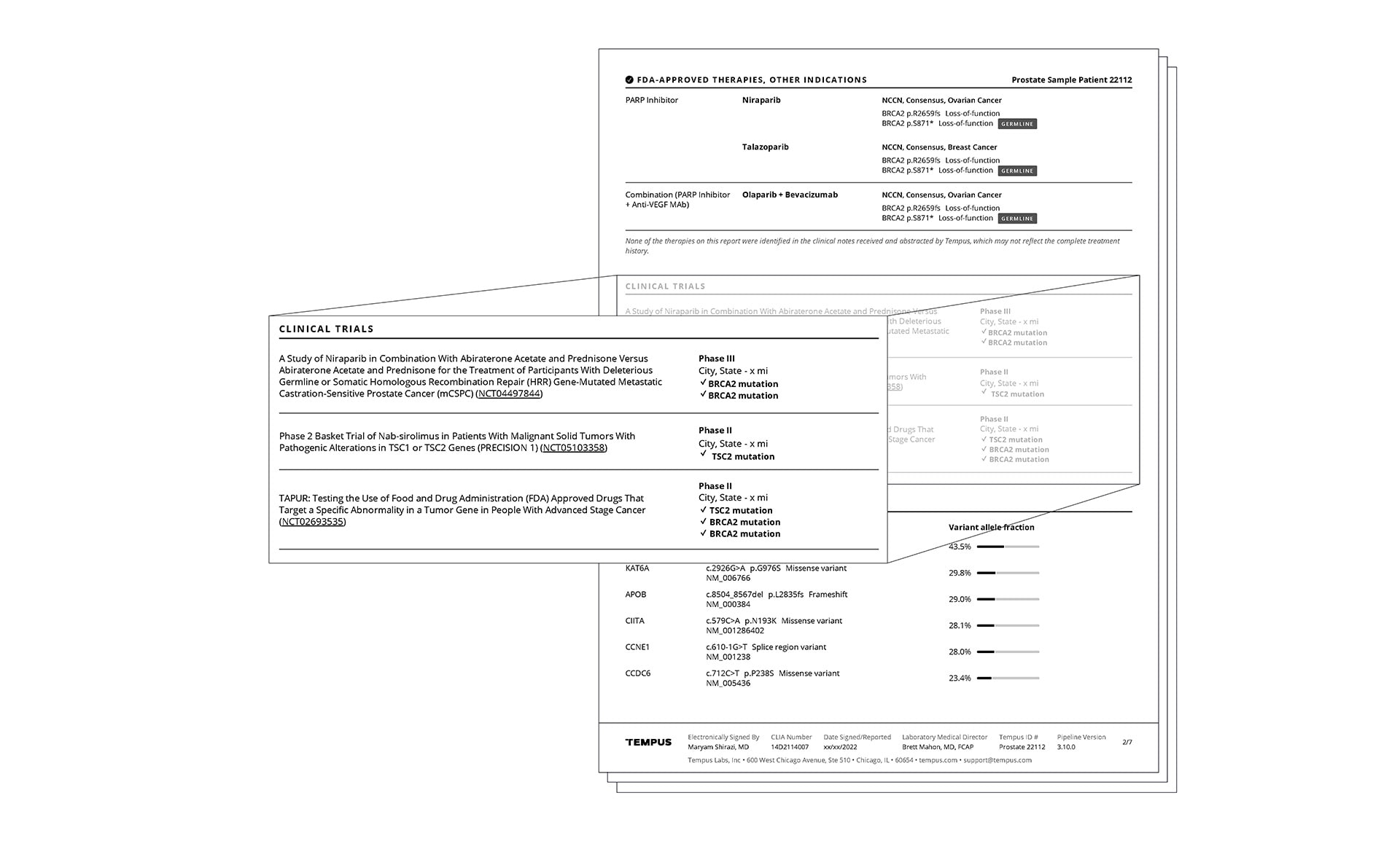
Actionable molecular findings.
-
PROVIDERS
Learn more
Join Tempus at the 2026 ASCO® Annual Meeting!
-
LIFE SCIENCES
Read case study
Learn now Tempus Next Pathways unlocked critical insights into the aNSCLC patient journey with Bristol Myers Squibb (BMS)
-
PATIENTS
It's About Time
View the Tempus vision.
- RESOURCES
-
ABOUT US
View Job Postings
We’re looking for people who can change the world.
- INVESTORS