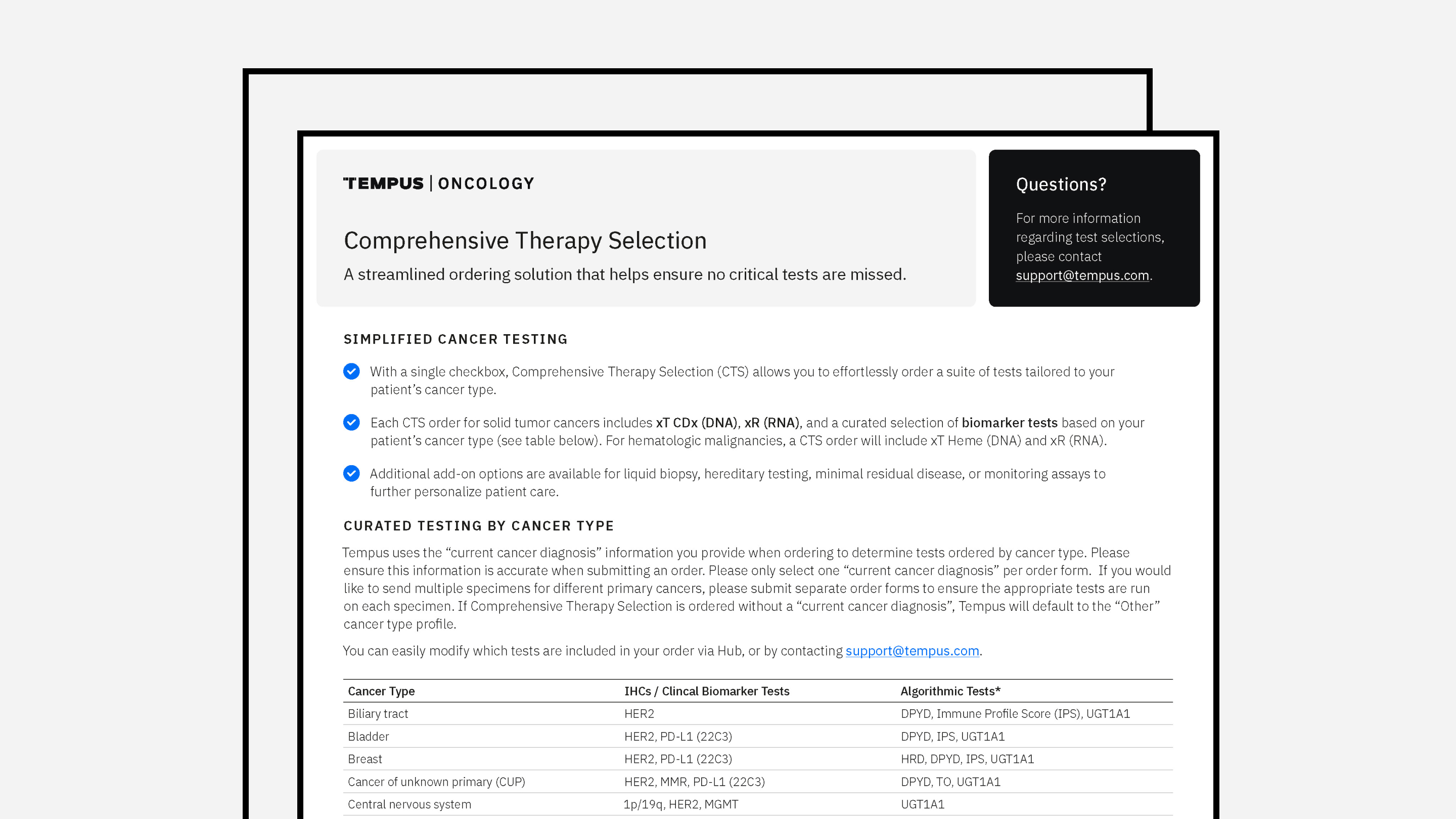
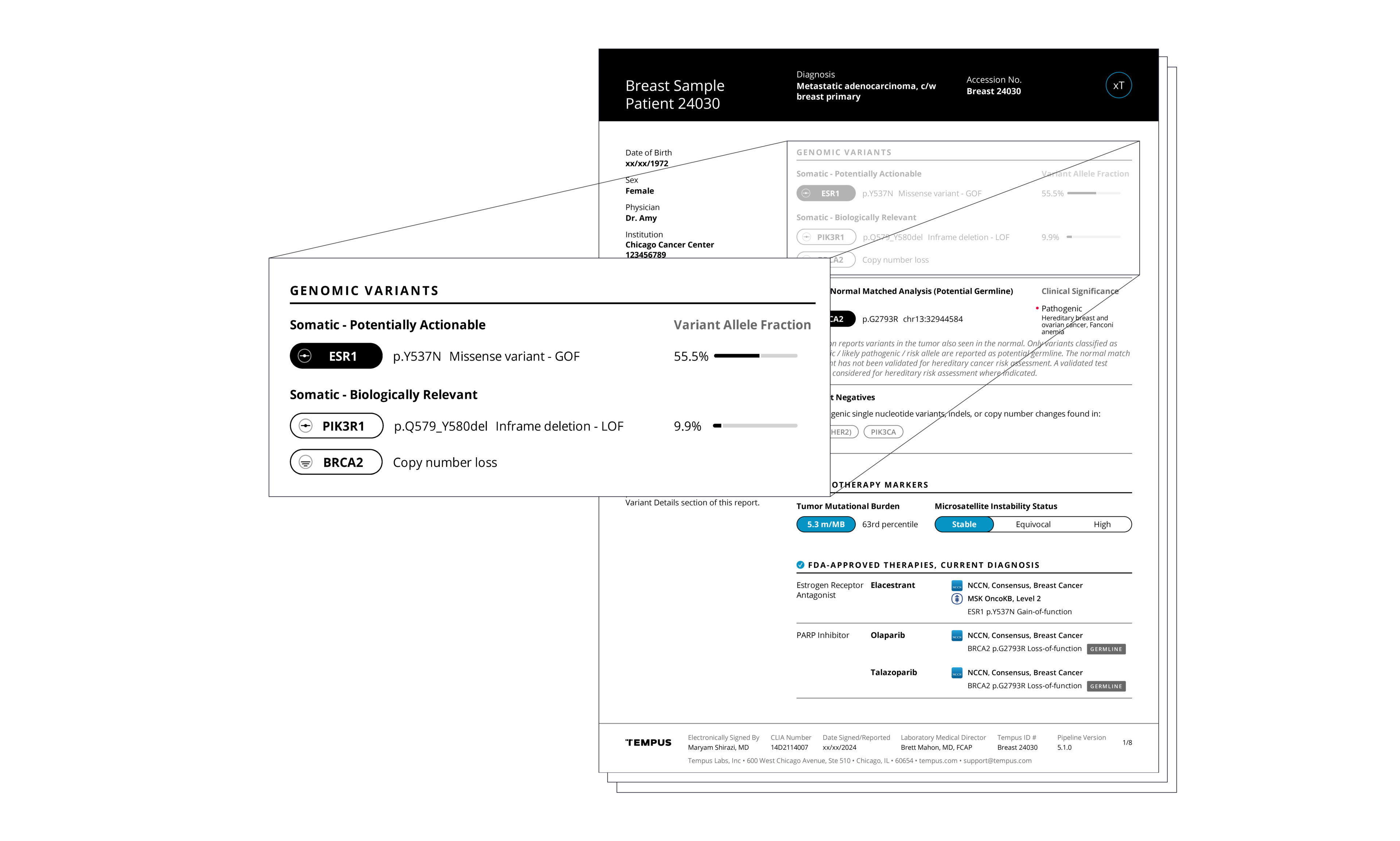
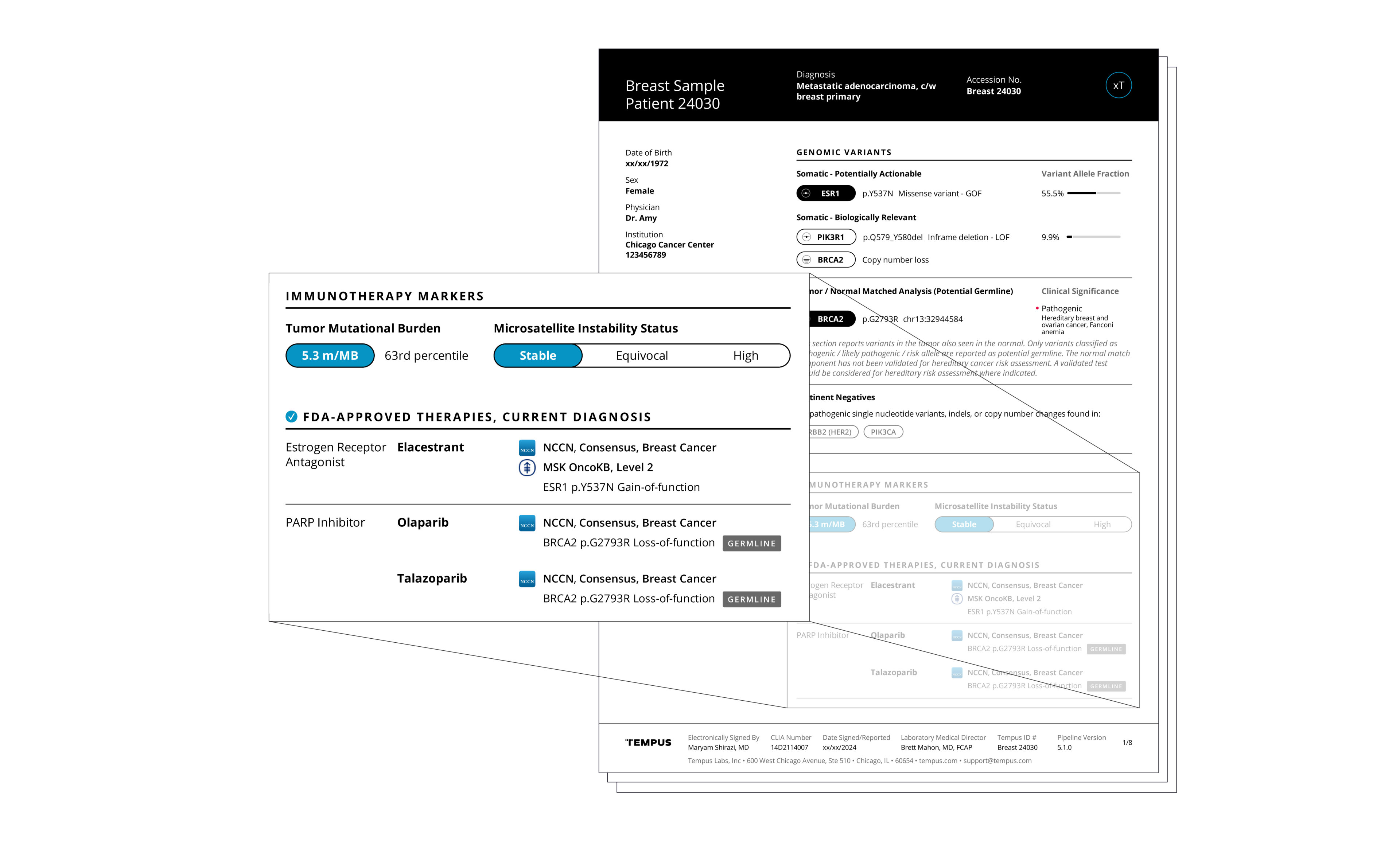
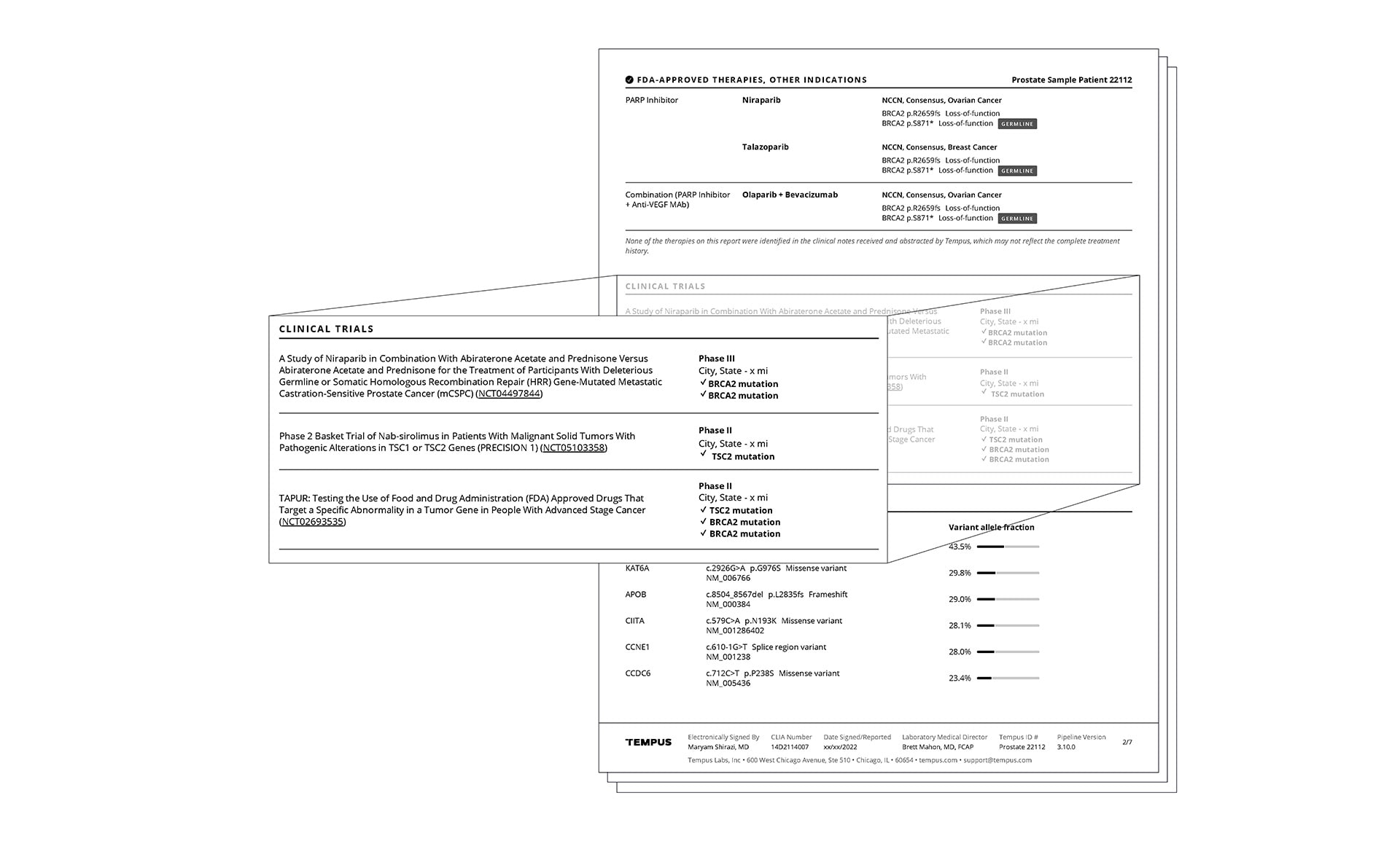
Actionable molecular findings.
-
PROVIDERS
REGISTER NOW
Upcoming Webinar:
MRD data insights across the cancer care continuum -
LIFE SCIENCES
REGISTER NOW
From insight to impact: Leveraging the AI-enabled Next platform with BMS to advance equitable access in precision oncology
-
PATIENTS
It's About Time
View the Tempus vision.
- RESOURCES
-
ABOUT US
View Job Postings
We’re looking for people who can change the world.
- INVESTORS